Methicillin resistant Staphylococcus aureus (MRSA) has been recognized as an important pathogen in nosocomial settings for many years. More recently, serious methicillin resistant S aureus infections from the community have been described in children in Minnesota and North Dakota who have died from these infections in 1997, 1998 and 1999 (Herold, 1998; MMWR, 1999). The children were noted to have matching strains of bacteria despite having no epidemiologic links and no hospital exposure. Since these initial reports, several groups have reported outbreaks of MRSA infections occurring outside of healthcare facilities, involving athletes, military personnel, and inmates in correctional facilities (Lindenmayer, 1998; MMWR, 2003a,b; Pan, 2003; Zinderman, 2004; Kazakova, 2005; Nguyen, 2005; Aiello, 2006) leading to the term community-acquired MRSA (CA-MRSA). CA-MRSA outbreaks in men who have sex with men (MSM) have been recently reported in several US cities, possibly associated with methamphetamine use and risky sexual behavior (Lee, 2005). This review summarizes the current knowledge of the epidemiology, clinical manifestations, diagnosis, and management of CA-MRSA infections with an emphasis on the HIV patient.
| Background | Top of page |
Since the early 1960's, MRSA strains have spread around the world causing nosocomial infections and outbreaks. These highly drug-resistant strains are distinguished into 5 major MRSA lineages and are further delineated based on the genetic organization of the staphylococcal chromosomal cassette that harbors the methicillin resistance determinant, mecA (SCCmec). In contrast to these highly drug resistant hospital strains, the Midwestern CA-MRSA strain (referred to as MW2) usually carries less antibiotic resistance determinants and is often sensitive to non-b-lactam agents (ie, clindamycin and aminoglycosides). Sequencing results have revealed MW2 to harbor a novel SCCmec type, referred to as SCCmec type IV (Kuroda, 2001; Hiramatsu, 2001), and this new promiscuous element has been found increasingly in S aureus strains. Since this observation, SCCmec type IV has rapidly evolved and numerous variants (SCCmec types IVa, IVb, IVc and V) have been described (Shore, 2005).
| Mechanism | Top of page |
What distinguishes methicillin-susceptible S aureus (MSSA) and MRSA is the acquisition of the staphylococcal cassette chromosome harboring the mecA gene. Expression of mecA yields a penicillin binding protein (PBP2a) that reduces the bacterium's susceptibility to all b-lactam drugs. It is interesting to note that the cassette that harbors the mecA gene is also capable of trapping drug resistant plasmids and transposons and thereby increasing the drug resistance of these S aureus strains. Through this mechanism, hospital-acquired MRSA (HA-MRSA) strains, and increasingly CA-MRSA strains, have become progressively resistant to a number of antibiotic classes. In addition to harboring SCCmec type IV, the Midwestern strain produces a toxin, Panton-Valentine leukocidin (PVL) that can destroy or lyse white blood cells (Daum, 2002). It is suspected that this leukocidin, which may facilitate skin infections, might have contributed to deaths of the children in the Midwest. Nearly all CA-MRSA strains appear to have the PVL gene, which has served as a genetic marker for identifying CA-MRSA.
| Hospital-Acquired Versus Community-Acquired Methicillin-ResistantStaphylococcus aureus | Top of page |
The US Centers for Disease Control and Prevention (CDC) has established a case definition for CA-MRSA, stating that the diagnosis must be made through a positive culture showing MRSA in an outpatient setting or within 48 hours of admission to the hospital (CDC, 2003). The patient must not have experienced any of the following during the year before infection: hospitalization; admission to a nursing home, skilled nursing facility or hospice; dialysis; or surgery. In addition, the patient must be without permanent indwelling catheters or medical devices that pass through the skin into the body (CDC, 2003). Genetic markers and virulence factors are useful in differentiating HA and CA infections; however, the difference between HA- and CA-MRSA is blurring with hospital strains being identified in the community and vice versa (Saiman, 2003; CDC, 2006).
Clinically, CA-MRSA differs from HA-MRSA in that it often occurs in diverse and otherwise healthy populations without clear common risk factors, such as young children, Native Americans, and MSM. The modes of transmission and the reservoirs have not been fully elucidated; however, infections are thought to be associated with skin-to-skin contact. Occasionally, CA-MRSA infections have been transmitted via fomites such as towels, public bath use, and sports equipment (Kazakova, 2005; Lee, 2005). In addition, reports of transmission via household pets have been noted (Manian, 2003; Strommenger, 2006).
| Epidemiology of Community-Acquired Methicillin-ResistantStaphylococcus aureus: Selected Cases | Top of page |
One of the first clearly identified community-acquired outbreaks of MRSA occurred in a Native-American population in Wisconsin (Stemper, 2004). A particularly large-scale outbreak occurred in the Los Angeles County Jail involving 928 inmates who were initially misdiagnosed as having spider bites (CDC, 2003a). Ten patients developed invasive disease, including bacteremia, endocarditis, and osteomyelitis.
From August to September of 2003, an outbreak of skin and soft tissue infections (SSTIs) due to CA-MRSA was identified among college football players in southern California (Nguyen, 2005). Eleven players were infected, most presented with boils. Linemen had the highest attack rate. Among 99 (93%) of the 107 players who had a culture of the nares performed, 8 (8%) were positive for MRSA. All MRSA isolates were genetically identified as MW2 (now classified as USA300). Risk factors for infection identified through a case-control study included sharing bars of soap and having preexisting cuts or abrasions. Having a locker near a teammate with an SSTI, sharing towels, and living on campus were associated with nasal carriage. Daily hexachlorophene showers and hygiene education appeared to stop transmission and end the outbreak.
In an outbreak that occurred during the 2003 season among members of a professional football team, 8 MRSA infections were reported among 5 (9%) of the 58 St Louis Rams players; all of the infections developed at turf-abrasion sites. Risk factors for a MRSA infection were the position of a lineman or linebacker and a higher body-mass index. No MRSA isolates were found in nasal or environmental samples. However, MSSA isolates were recovered from whirlpools, taping gel and from 35 of 84 (42%) of nasal swabs from players and staff (Kazakova, 2005). Isolates of MRSA from infected team members and from a competing football team were identified as USA300 and carried the PVL gene.
| MSM and HIV-Infected Persons | Top of page |
S aureus is not considered an opportunistic infection, but several studies have found an association between HIV and MRSA. Researchers at the University of California, San Diego investigated trends in the incidence of, and risk factors for, clinically significant MRSA infections among 3,455 HIV-infected adults seen in their clinic (Mathews, 2005). From 2000 through 2003, MRSA-positive clinical isolates increased 6.2-fold from the first to the last half of the study; 60% of the MRSA isolates were determined to have been community-acquired by epidemiologic characteristics. Risk factors associated with CA-MRSA infections among HIV patients included a history of MSM, injection drug use, or both as patients' risk factors for HIV acquisition (Mathews, 2005). In addition, high HIV viral load and CD4+ cell counts less than 50 cells/mm3 were predictors of CA-MRSA infections. Prophylaxis with trimethoprim-sulfamethoxazole (TMP-SMX) for 120 days or more was found to be protective of CA-MRSA infections (P<.005) (Mathews, 2005).
In a recent retrospective study from 1993 to 2005 of CA-MRSA infections among 425 HIV patients in San Diego, California all CA-MRSA cases occurred after 2002, with a 17-fold increase in the number of CA-MRSA infections from 2003 through 2005 (Crum-Cianflone, 2006). Not only did the number of cases rise over time, but the percentage of S aureus infections due to CA-MRSA compared with MSSA also increased; by 2004 through 2005, 65% of all staphylococcal SSTIs were due to CA-MRSA in this cohort.
In summary, these studies and others suggest that poor HIV control, antibiotic usage, and perhaps high sexual risk behaviors may place HIV patients at heightened risk for CA-MRSA infections. In addition, some studies suggest that use of public hot tubs or saunas increased the risk of CA-MRSA (Lee, 2005), hence avoiding these locations may reduce CA-MRSA infections among HIV-infected persons. Furthermore, these retrospective studies among HIV-infected patients demonstrate that the rate of CA-MRSA infections are rapidly increasing and should be suspected among HIV patients and those with high-risk behaviors presenting with SSTIs (Table 1) (Iyer, 2004). TMP-SMX, when used for Pneumocystis pneumonia (PCP) prophylaxis in AIDS, also appears to be very effective at preventing MRSA infections; however, this trend could change if CA-MRSA develops resistance to this antibiotic.
| Table 1. Summary of Studies of Community-Acquired Methicillin-Resistant Staphyloccus Aureus Among HIV-Positive Persons | |||
| Study | Epidemiologic Trends | Risk Factors for MRSA | Protective Factors against MRSA |
| Mathews, 2005 | 6.2-fold increase in cases from 2000–2003 among an HIV cohort at the Owen Clinic, San Diego | • MSM, IDU, or both as HIV transmission risk factors |
• TMP/SMX use |
| Lee, 2005 | Cross-sectional study in MSM | • Public bath/sauna use • Methamphetamine use • Sexual partner with a skin infection |
• TMP/SMX use • Condom use |
| Crum-Cianflone, 2006 | 17-fold increase in cases from 2003–2005 among a HIV cohort in San Diego | • Recent use of β-lactam antibiotics • History of syphilis • Low current cd4 count • High maximum viral load |
• TMP/SMX use* |
| * Did not reach statistical significance in this study, but appeared to be protective. | |||
| Recent Epidemiologic Investigations in the Community | Top of page |
At the meeting of the Physicians' Research Network in New York City on January 17, 2006, Dr. Barry Kreiswirth of the Public Health Research Institute (PHRI) presented results from a recent surveillance of CA-MRSA in New Jersey. The goals of the study were to determine the frequency of MRSA and its genotypes by capturing all S aureus isolates submitted to a large commercial laboratory, Quest Diagnostics, from the offices of northern New Jersey physicians in private practice. The researchers also obtained specific patient information via a questionnaire sent to the patients' doctor.
Of more than 1000 S aureus isolates, collected from both male and female patients between the ages of 2 weeks and 99 years, 28% were identified as MRSA strains. Nearly all were from skin infections, and 80% of the cases had no recent connection to a hospital (Barry Kreiswirth, unpublished data). The MRSA strains were categorized into 2 groups: those that were SCCmec type II and PVL negative, presumed to be HA-MRSA (n=124), and those that were SCCmec type IV and PVL positive, presumed to be CA-MRSA (n=99). Since SCCmec type II has been identified as a hospital clone, and all the samples in this study came from patients at physicians' offices, nosocomial infections may be causing new skin infections in the community.
In July of 2005, the New York City Department of Health and Mental Hygiene (NYCDOHMH) began an epidemiologic investigation to determine the incidence and risk factors for patients with CA-MRSA skin infections diagnosed in outpatient settings in New York City. The ultimate goal of the investigation is to design an intervention targeted at high-risk groups to interrupt CA-MRSA transmission. For this investigation, NYCDOHMH receives isolates and reports which include susceptibility to various antibiotics, from all patients diagnosed with MSSA and MRSA by a large outpatient laboratory. Then, these isolates are genotyped, spa typed, and tested for PVL by public health laboratories and PHRI. Patients are called by phone, and interviewed with a structured questionnaire to ascertain demographics and risk factors for CA-MRSA. Whether the patient's infection was community-acquired or hospital-acquired is determined by epidemiologic criteria. Infections are defined as community-acquired if a patient denied having stayed overnight in a hospital and having had dialysis in the 3 months prior to his/her symptom onset.
According to the laboratory records of the first 99 patients interviewed, 88% were resistant to erythromycin, 73% to ciprofloxacin, 71% to levofloxacin, and 45% to clindamycin (includes D-test). Only 22% of isolates were resistant to tetracycline, while rifampin and trimethoprim-sulfamethoxazole resistance was low at 3% and 2%, respectively.
There were differences in antibiotic susceptibilities across age groups. For example, resistance to fluoroquinolones among patients aged 0 to 17 years was 39% compared with 73% among patients between 18 and 64 years of age and 90% in patients 65 years or older. In addition, multidrug resistance was fairly common with only 6% of the isolates sensitive to all 6 classes of potentially effective antimicrobial drugs; 78% were resistant to 2 or more drugs; 46 % to 3 or more; and 12% to 4 or more.
Among MRSA-infected patients from a larger sample (N=458) of the same NYC study, 64% were men, the mean age was 44 years, and eighty four percent (N=382) of infections were classified as community-acquired. Of the 382 CA-MRSA patients interviewed, 67% were men, and 70% of men reported having had sex with a man in the prior 12 months (MSM), which is 7 times the rate in the general population of NYC men (10%). Twenty-seven percent of CA-MRSA infected men reported being infected with HIV, which is 27 times the rate in men in the general New York City population.
The men who had sex with men in this sample reported "having ever used" the following drugs: sildenafil (Viagra®) (29%), methamphetamine (24%), ecstasy (21%), cocaine (18%), heroin (2%) and crack (1%). MSM interviewed also reported a median number of 2 sex partners in the prior 3 months, but responses to this question varied considerably (range 0-40). They reported having had anal receptive intercourse 4 times, but having used a condom only (a median of) once in the 3 months prior to the interview. The low frequency of condom use can not be evaluated as a risk factor for MRSA transmission until controls are included in the analysis, which will not occur until the second phase of the investigation.
Of the initial group studied, the majority of patients were treated with antibiotics (96%) and a large proportion with incision and drainage (58%).
When the first phase of the investigation is completed, controls will be interviewed to compare risk factors. After data are analyzed and risk factors identified, the NYCDOHMH will release guidelines for preventing CA-MRSA to the public and summaries of antibiotic-susceptibility profiles to clinicians to aid treatment decisions.
A third community-based investigation which examined patients presenting with skin and soft tissue infections to the emergency room was recently published (Moran, 2006). S aureus was the cause of the infection in 76% of cases, 59% of which were due to MRSA. Of the MRSA isolates, 97% were type USA 300, the most common isolate associated with community-acquired strains. This study clearly demonstrates that CA-MRSA is the most common microbiologic cause of skin and soft tissues infections occurring in the community.
| Clinical Manifestations of Community-Acquired Methicillin-Resistant Staphylococcus aureus Infections | Top of page |
The most common clinical presentation of CA-MRSA skin or soft tissue infection is an abscess with or without a surrounding cellulitis (Figure 1). Common clinical presentations include furuncles (Figure 2), carbuncles, and folliculitis (Figure 3).
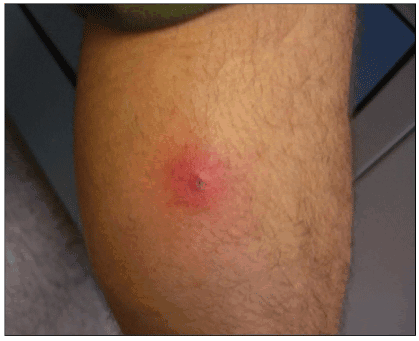
Figure 1. Classic CA-MRSA soft tissue infection (furuncle)
Photo by Peter Ruane
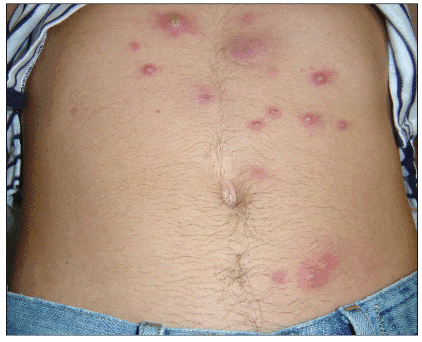
Figure 2. Furunculosis
Available at: http://www.DermAtlas.org.
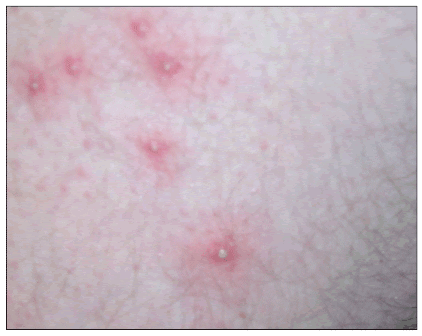
Figure 3. Folliculitis
Available at: http://www.DermAtlas.org.
The furuncles and abscesses of CA-MRSA infections frequently produce a brisk erythematous response in the surrounding tissue a characteristic appearance which is likely related to PVL and other pathogenic factors (Figure 4) and can rapidly progress in size (Figure 5). Early central necrosis may be present and the lesions may be mistaken for spider or other insect bites (Figure 6).
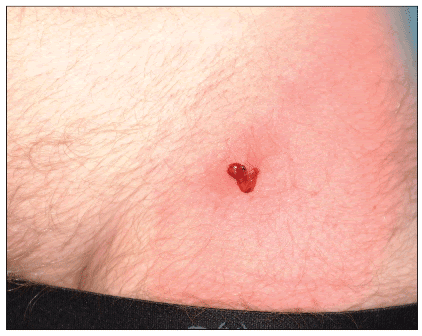
Figure 4. Painful left groin abscess with pinpoint discharge of hemopurulent material and extensive cellulitis
Photo by Peter Ruane
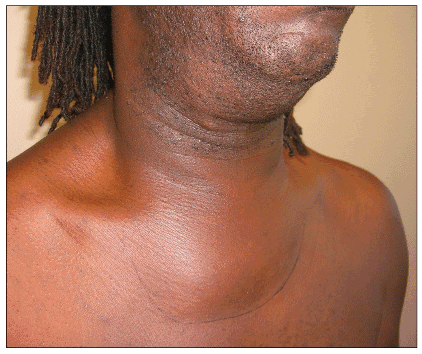
Figure 5. Extensive involvement of MRSA infection in the soft tissue of the neck
Infections around the head and neck may be associated with shaving practices as well as nasal carriage of the organism./p>
Photo by Peter Ruane
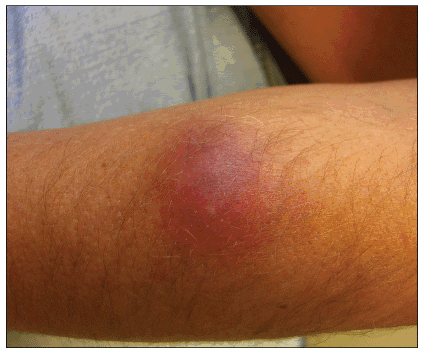
Figure 6. Intensive cellulitis of the upper extremity with CA-MRSA
Photo by Peter Ruane
Recurrence of furuncles or abscesses may occur weeks to months after the initial CA-MRSA infection (Figure 7) despite appropriate treatment of the first infection. Recurrence may be related to persistence of the organism in the nares or other warm moist body sites, or due to reinfection from an outside source. Recurrence rates vary; Crum et al reported a 9% recurrence rate (Crum, 2006), but recurrence rates may be higher in high-risk populations including HIV patients (Crum-Cianflone, 2006).
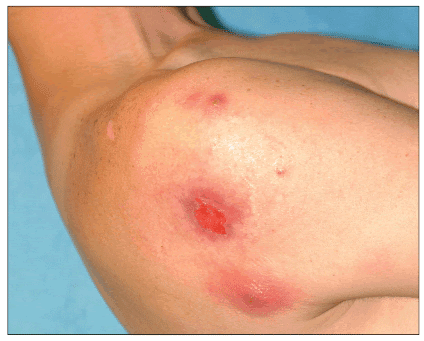
Figure 7. Abscess with ulceration in patient with recurrent MRSA
Photo by Peter Ruane
Less common SSTIs include impetigo (Figure 8), paronychia (Figure 9), and cellulitis without abscess. Impetigo may be localized or extensive, and is characterized by diffuse yellow honey-like crusting. Paronychia may initially present as slight edema of the proximal or lateral nail fold with some redness or pain, but can develop into a frank abscess or pustule. Occasionally CA-MRSA may cause cellulitis without an associated furuncle or abscess (Figure 10) in the absence of a culture, CA-MRSA might be suspected if the infection fails to respond to initial treatment with dicloxacillin or a cephalosporin.
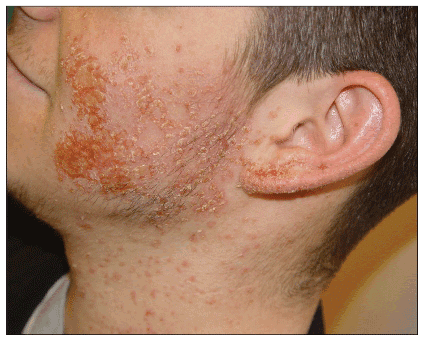
Figure 8. Impetigo
Available at:www.clinical-virology.org/gallery
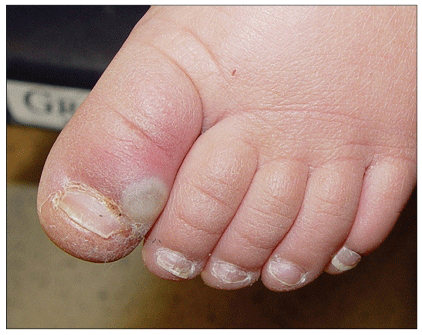
Figure 9. Paronychia
Available at: http://www.DermAtlas.org
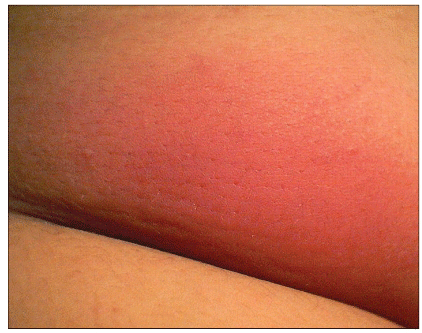
Figure 10. Cellulitis
Available at: http://www.DermAtlas.org
Although CA-MRSA soft tissue and skin infections may occur at any body site, studies among MSM and/or HIV patients report a predominance of these lesions on the buttocks and scrotal areas (Iyer, 2004; Crum-Cianflone, 2006). At a West Hollywood California dermatology clinic, most common sites of MRSA infection were the genital area and buttocks (21.7%) as well as the face and neck (21.7%) (Iyer, 2004). The next most common sites included the legs (17.4%) and the back (10.9%).
Other important but less common complications of CA-MRSA include pyomyositis (Fowler, 2006), necrotizing fasciitis (Figure 11) (Miller, 2005), osteomyelitis and septic arthritis (Fridkin, 2005). Necrotizing pneumonia has been described; cases primarily occurred in children, but older age groups may be affected (Francis, 2005). CA-MRSA bacteremia, toxic shock, and endocarditis among otherwise healthy persons have been reported (Gonzalez, 2005; Mongkolrattanothai, 2003).
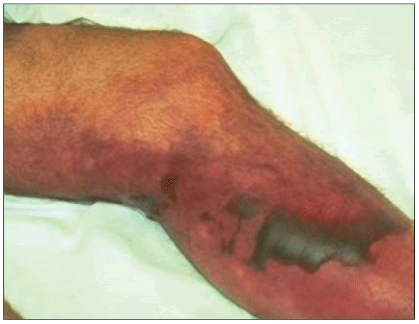
Figure 11. Necrotizing Fasciitis
Available at:euclid.dne.wvfibernet.net
It is important that MRSA be considered in the differential diagnosis of SSTIs and other syndromes compatible with S aureus infection.(Table 2).
The definitive diagnosis of CA-MRSA SSTIs relies upon the recovery in culture of the organism from the lesion. Among patients with skin infections that cannot be cultured (eg, cellulitis), a culture from another site, for example the nares or other warm areas such as the perineum or perirectal area may confirm carriage of MRSA.
| Table 2. Clinical Manifestations of Community-Acquired Methicillin-Resistant Staphyloccus Aureus Infections |
| Skin/Soft Tissue Infections |
| • Abscess • Furuncle • Carbuncle • Folliculitis • Impetigo • Paronychia • Cellulitis (usually in association with one of the above soft tissue infections) • Wound infection |
| Other Clinical Manifestations |
| • Pyomyositis (abscess in a large skeletal muscle) • Necrotizing fasciitis • Bone infection (osteomyelitis) • Joint infections (septic arthritis) • Bacteremia • Toxic shock syndrome • Endocarditis • Necrotizing pneumonia • Any type of infection |
| Treatment | Top of page |
There are currently no randomized clinical trials which establish the optimal antimicrobial therapy for CA-MRSA infections. Treatment guidelines for SSTIs that address the rising rate of CA-MRSA as the etiologic agent of these infections are available at the CDC Web site (http://www.cdc.gov/ncidod/dhqp/pdf/ar/CAMRSA_ExpMtgStrategies.pdf) as well as via a publication by the Infectious Disease Society of America (IDSA) (Stevens, 2005), and from other groups (http://www.metrokc.gov/health/providers/epidemiology/MRSA-guidelines.pdf). While neither the CDC nor IDSA provide specific recommendations for empiric therapy against CA-MRSA when treating all SSTIs, many clinicians initiate antibiotics effective against CA-MRSA especially in cases of severe infection or toxemia (King, 2006). In addition, some physicians initiate therapy with agents active against CA-MRSA in patients at risk (such as those with a prior history of CA-MRSA or HIV-infected persons) who have typical lesions.
| Table 3. Clinical Manifestations of Community-Acquired Methicillin-Resistant Staphyloccus Aureus Infections | |
| Antibiotics which are typically effective against CA-MRSA* | Antibiotics which are usually ineffective against CA-MRSA* |
| • Tetracyclines • Doxycycline (Vibramycin®) • Minocycline (Minocin®) • Trimethoprimsulfamethoxazole (Bactrim®, Septra®) • Rifampin (Rifadin®) • Clindamycin (Cleocin®)** • Vancomycin (Vancocyn®) • Linezolid (Zyvox®) • Daptomycin (Cubicin®) • Tigecycline (Tygacil®) • Quinupristin/dalfopristin (Synercid®) • Topical mupirocin (Bactroban®) |
• All penicillin agents
(always ineffective) • All cephalosporins (always ineffective) • All carbapenems • Erythromycin (E-mycin®) • Flouroquinolones |
| *Susceptibility must be confirmed by the microbiology laboratory. **Susceptibility to clindamycin should be confirmed with D-testing. |
|
Table 3 lists antimicrobial agents considered for the treatment of CA-MRSA. Choice of antibiotics in the treatment of CA-MRSA infection is determined by the susceptibility of the organism and the severity of the infection. CA-MRSA strains are resistant to all penicillins and all cephalosporins. Isolates are often resistant to erythromycin, and only variably susceptible to the fluorquinolones (Fridkin, 2005), hence these agents should not be used without performing susceptibility testing. Most CA-MRSA isolates remain sensitive to clindamycin (Moran, 2006; Fridkin, 2005); however, increasing rates of resistance to clindamycin have been reported. (Braun, 2005). Isolates that are clindamycin susceptible but erythromycin resistant during the initial antibiotic susceptibility testing, should be confirmed to be susceptible to clindamycin with the D test which assesses for inducible resistance (Lewis 2005). If inducible resistance is detected by the D test, clindamycin is reported as resistant and should not be considered for therapy (Siberry, 2003; Lewis, 2005).
A recent study of 1,647 cases of CA-MRSA infections from 3 communities (Baltimore, Atlanta, Minnesota) showed that all CA-MRSA isolates were susceptible to vancomycin, 97% to trimethoprim-sulfamethoxazole, 98% to rifampin, 96% to linezolid, 88% to tetracyclines, 87% to clindamycin, 65% to ciprofloxacin, and 18% to erythromycin (Fridkin, 2005). Similar antibiotic resistance rates were found among patients presenting to university-affiliated emergency rooms in large US cities for a skin or soft tissue infection (Moran, 2006). Clinicians should be aware of local susceptibility patterns to optimize selection of best empiric therapy. Based on the study by Fridkin and colleagues as well as other data, Table 3 presents agents to avoid and agents to consider using for CA-MRSA infections.
| Antimicrobial Therapy of CA-MRSA | Top of page |
Patients who are severely ill or have a rapidly progressing infection should be referred to the hospital for consideration of intravenous antibiotics. Intravenous vancomycin is the most commonly used antibiotic for this indication. Emergence of vancomycin-intermediate and vancomycin-resistant MRSA (VISA and VRSA, respectively) has been reported, but are uncommon (Appelbaum, 2006). Linezolid may offer an alternative to intravenous vancomycin. Recent studies have shown that the adverse event rate of linezolid is not significantly different than that for vancomycin (Stevens, 2002) and that linezolid is an effective agent for SSTIs including those caused by MRSA (Weigelt, 2005). Daptomycin has been used effectively in cases of complicated SSTI and for treatment of CA-MRSA bacteremia (Arbeit, 2004). Quinupristin/dalfopristin is active against MRSA, but is rarely used due to an adverse-effect profile and potential cross-resistance with clindamycin-resistant strains. Tigecycline is active against MRSA and it has FDA approval for the treatment of skin and soft tissue infections (Ellis-Groose, 2005).
Patients with localized infection and without systemic toxicity may be managed as outpatients with oral medications. Oral antibiotic therapy should be continued until there is resolution of signs of acute inflammation; this typically occurs within 7-14 days (Stevens 2005; Swartz 2005). Options for oral antibiotics include trimethoprim-sulfamethoxazole, clindamycin, linezolid or tetracyclines (eg, minocycline or doxycycline ) (Table 4) (Moran, 2006; Lewis, 2005; Rybak, 2005; Johnson, 2006). Rifampin is also usually highly active against CA-MRSA and penetrates both tissues and mucosal surfaces well and can be combined with one of these antibiotics. Rifampin when used alone is associated with rapid emergence of resistance and should not be used in this manner.
Access to oral linezolid may be limited due to formulary restrictions and other cost related issues. Some infectious disease specialists save linezolid for use in infections due to organisms resistant to other agents. This conservative approach is supported by reports of the emergence of linezolid-resistant organisms in healthcare settings (Kelly, 2006).
| Table 4. Commonly Used Drugs and Their Dosages for Community-Acquired Methicillin-Resistant Staphyloccus Aureus Infections* |
| Intravenous Antibiotics |
| • Vancomycin (Vancocyn®) 15 mg/kg IV q12 hours • Daptomycin (Cubicin®) 4 mg/kg IV daily (higher dosages are used for bacteremia/endocarditis) |
| Intravenous or Oral Antibiotics |
| • Linezolid (Zyvox®) 600 mg IV or PO twice daily • Clindamycin (Cleocin®) 900 mg IV q8 hours or 300–450 mg PO QID |
| Oral Antibiotics |
| • Tetracyclines • Doxycycline (Vibramycin®) 100 mg PO twice daily • Minocycline (Minocin®) 100 mg PO twice daily • Trimethoprim-sulfamethoxazole (Bactrim®, Septra®) 1 double-strength with 160 TMP/800 SMX tablet twice daily • Rifampin (Rifadin®) 300 mg PO twice daily |
| Topical Antibiotics |
| • Topical mupirocin (Bactroban®) apply to each nares twice daily • Chlorhexidine body soaps, shower with soap daily (used for decolonization purposes, not treatment) |
| * Dosages listed assume normal kidney and liver function; for patients with abnormal values, drug dosage adjustments may be needed. Some antibiotics listed are not recommended in children or during pregnancy. |
Topical, rather than oral, antibiotics can be used to treat superficial lesions. For instance, topical mupirocin TID for ~7 days has been utilized for treatment of limited impetigo (Stevens, 2005; Swartz 2005). Of note, resistance to mupirocin may develop, but this usually occurs in the setting of prolonged usage (Yun, 2003). For multiple or larger CA-MRSA lesions, oral antibiotics are recommended.
| Methicillin-Resistant Staphylococcus aureusTreatment in the Setting of Highly Active Antiretroviral Therapy | Top of page |
Although several oral agents are available, co-infection with HIV and the use of antiretroviral therapy may limit treatment options in some patients. TMP-SMX use may be limited in HIV patients due to pre-existing drug allergy. Clindamycin may be associated with Clostridium difficile colitis, a potentially vexing issue among HIV-patients (Sanchez, 2005). Linezolid use may be limited by thrombocytopenia necessitating weekly monitoring of complete blood counts. Linezolid may also interact with psychotropic medications such as monoamine oxidase inhibitors (MAOIs) and selective serotonin reuptake inhibitors (SSRIs). Ideally patients should be titrated off of SSRIs prior to taking linezolid, but in many clinical situations this may not be possible. In those situations, patients should be warned to watch for and report any signs or symptoms suggestive of the serotonin syndrome which include hyperthermia, agitation, tremors, myoclonus, altered mental status, and/or diaphoresis (Taylor, 2006).
Drug-drug interactions limit the co-administration of rifampin with several antiretroviral agents including protease inhibitors and non-nucleoside reverse transcriptase inhibitors. Rifampin is a powerful inducer of the cytochrome P450 enzyme system, leading to a decrease in antiretroviral plasma levels below the inhibitory concentration of 50% (IC50) of the latter and potentially leading to viral rebound. Rifampin should be avoided in patients receiving protease inhibitors (Niemi, 2003) and an alternate antibiotic, such as clindamycin, linezolid or tetracycline should be selected.
| Recurrence of CA-MRSA Infections: | Top of page |
Results from an HIV cohort in San Diego, California (Crum-Cianflone, 2006) demonstrated high rates of recurrent CA-MRSA infections. Patients with recurrent infections despite adequate initial therapy should be considered for decolonization treatment. A recent study among military trainees showed benefit of decolonization (Ellis, 2004), but this study was conducted in an immunocompetent patient population. Swabs of the nares, axilla, groin, perineum and pharynx (Ringberg, 2006; Stevens, 2005) have been utilized to establish carriage. Follow-up surveillance cultures may be obtained to ensure clearance after therapy, but recolonization may occur despite these results (Marschall, 2006). At this time, routine surveillance cultures to screen for colonization with CA-MRSA after completion of treatment have not been recommended by the CDC or other scientific groups.
Although optimal regimens for decolonization have not yet been established by clinical trials, most clinicians use topical agents including intranasal topical mupirocin administered bid for 5-7 days along with chlorhexidine body wash daily for 5-7 days (Boyce, 2001; Sandri, 2006). Although mupirocin resistance is described (Hurdle, 2005), the rate of resistance appears low. Other agents for decolonization include a variety of oral antibiotics such as rifampin combined with trimethoprim-sulfamethoxazole, minocycline or doxycycline.
Surgical Treatment Methods
Incision and drainage of an abscess may be effective treatment without concurrent antimicrobial agents. Some authors have suggested this approach may be utilized in health patients with no systemic signs of infection, who present with a small abscess without concurrent cellulitis. (Fridkin, 2005; Moran, 2006). Furuncles and abscesses require drainage. While this can spontaneously occur, many cases require incision and drainage. Multiloculated abscesses are common and may require aggressive exploration and drainage. For small to moderate sized purulent collections, incision and drainage can be performed within the office setting. Patients with deep soft tissue infections or necrotizing fasciitis should be referred for immediate surgical consultation.
Patient Education and Prevention
Patients often have many questions regarding how and why they developed a CA-MRSA infection. Explaining to the patient the epidemiology, diagnosis and management of these infections in layman's terms is important.
In addition, patients should be instructed regarding the following items in an attempt to prevent further spread of the organism to their contacts:
1. Keep draining wounds covered
2. Maintain good hygiene
3. Wash hands regularly and after touching the wound
4. Avoid sharing items such as towels, clothes, bars of soap, or razors
5. Avoid direct contact (e.g., playing contact sports) with others unless the wound can be covered
6. Wash thoroughly all clothes in contact with the wound site
7. Clean equipment or other environmental surfaces with antibacterial agents that have activity against S aureus
8. Shower with hot water and soap after exercising
Patients should be instructed that CA-MRSA infections may recur, and that new skin lesions should be promptly reported to their physician. In addition, contacts of the patient who develop a suspected MRSA infection should be referred. Guidelines for reducing the spread of CA-MRSA are available at http://lapublichealth.org/acd/MRSA.htm. Physicians should report suspected outbreaks of CA-MRSA infection that are occurring in groups of patients with potentially the same exposure to the public health department. This includes among athletic teams, households, schools, and so forth. In addition, increasing rates can be reported. Participation in surveillance studies is also valuable for the further understanding of epidemiologic trends for CA-MRSA infections.
| Conclusions | Top of page |
Clinicians should maintain a high level of suspicion for CA-MRSA among HIV patients with SSTIs. All suspicious soft tissue and wound infections should be cultured. Outpatient treatment includes TMP-SMX, clindamycin, tetracylines and linezolid. Intravenous agents include vancomycin, linezolid or daptomycin. Rifampin may be utilized in combination with any of the preceding oral or intravenous agents. Adequate drainage of purulent collections is required. Patients should be forewarned of the risk of recurrence. Studies to evaluate the efficacy of decolonization are inconclusive. Nevertheless decolonization of skin and mucosal surfaces is often attempted, particularly in cases of recurrence.
| References | Top of page |
